FDA flippantly authorizes Pfizer booster for 5 to 11-year-olds as agency capture hits breakaway speeds
The agency’s recent move shows the state of science in America is terminally ill.
The U.S. Food and Drug Administration (FDA) has green-lit another Pfizer Covid vaccine for children. This time around it's an Emergency Use Authorization (EUA) for a Pfizer booster aimed at kids 5 to 11 years of age – to make up for the lack of coverage from the failed 2-dose regime okayed in October of 2021.
In what has now become commonplace for the agency, the FDA can no longer be bothered to publicly discuss the science or data surrounding its vaccines receiving an EUA. Despite the FDA's budget for Fiscal Year 2021 of $6.1 billion, they could only muster a one-page press release to dictate to Americans that a third shot will now be coming to a kid's arm near you.
So what is the gold standard science contained in that press release?
For starters, the safety of the now authorized booster dose was assessed “in approximately 400 children” for a limited timeframe. Not looking to Sciency so far. The press release goes on to explain why, again, they didn’t convene a VRBPAC committee meeting to hold a vote on the matter.
“The FDA did not hold a meeting of its Vaccines and Related Biological Products Advisory Committee on today’s action, as the agency previously convened the committee for extensive discussions regarding the use of booster doses of COVID-19 vaccines and, after review of Pfizer’s EUA request, the FDA concluded that the request did not raise questions that would benefit from additional discussion by committee members.”
Got that! The agency looked at Pfizer’s EUA request and “concluded that the request did not raise questions that would benefit from additional discussion by committee members.” That’s Pfizer’s data…you know, the company whose vaccine product, is free from liability and poised to make countless millions in revenue. The same Pfizer who paid one of the biggest fraud settlements in U.S. history of $2.3B for false claims. That Pfizer.
The FDA has bypassed its ‘expert committee’ before without much commentary or fanfare. Yet, this time the agency decided to give its VRBPAC committee some good old-fashioned public humiliation insinuating that no benefit would come from their combined wisdom, experience, and knowledge…No questions, no debate, nothing.
The press release also underwhelms by stating that the efficacy, which couldn’t possibly be questioned by experts, looked at a measly 67 study participants.
“The antibody level against the SARS-CoV-2 virus one month after the booster dose was increased compared to before the booster dose.” states the FDA.
One month!?!? What happens after two months? We’ll just have to wait and see I guess.
And what is the reasoning for rushing a Covid booster for the 5-11 age group through so fast and recklessly?
According to the American Academy of Pediatrics (AAP) Covid-19 State-Level Data Report tracking cumulative hospitalizations in kids throughout 25 states (and NYC) since the start of the pandemic, “0.1%-1.5% of all their child COVID-19 cases resulted in hospitalization.”
As for cumulative mortality in 46 states (and NYC, PR, and GU) “In states reporting, 0.00%-0.02% of all child COVID-19 cases resulted in death.”
Meanwhile, the U.S. Centers For Disease Control and Prevention (CDC) MMWR report recently showed that seroprevalence of infection-induced SARS-CoV-2 antibodies, which are produced in response to infection but not in response to Covid-19 vaccines, found that from December 2021–February 2022, overall U.S. seroprevalence increased from 44.2% to 75.2% among children aged 0–11 years.
Since the FDA declined to bring in its experts for commentary or public vote, we are forced to look back to October 2021 when the agency gave its EUA to Pfizer’s 2-dose series for the same 5-11-year-old ages. At that meeting, the lack of evidence to approve Pfizer’s shot was clear. However it was Michael Kurilla, M.D., Ph.D., the only member who abstained from voting (while everyone else voted a unanimous ‘yes’), had this to say: [starts at 9:06]
Kurilla makes several important points. First, the correct assumption that natural immunity numbers are higher for the 5 to 11’s than is being reported. Second, does vaccinating these kids with an old (ancestral) strain even help, and for how long?
Outside the agencies, what are the medical journals saying? A new study from JAMA looked at Children (5-11) and Adolescents (12-15) who received Pfizer’s 2 dose Covid shots and their association with symptomatic SARS-CoV-2 Infection during omicron predominance.
The authors concluded:
“Among children and adolescents, estimated VE for 2 doses of BNT162b2 against symptomatic infection was modest and decreased rapidly.”
Looking at the graph from the study shows that the adjusted vaccine effectiveness dropped into the negative range around the 5-month mark for kids 12-15 years of age. And the 5-11’s hovered around 20% to 30% for months one and two.
In the world of Covid-boosting adults, much further along than kids at the moment, words like vaccine ceiling effect, immune exhaustion, immune tolerance, and booster fatigue have all hit the popular lexicon to deal with the results of continually vaccinating in the Covid era. There’s even the potential for a Covid vaccine-induced AIDS-like syndrome being noted as the immune system appears to down-regulate in the recipients of multiple Covid vaccines.
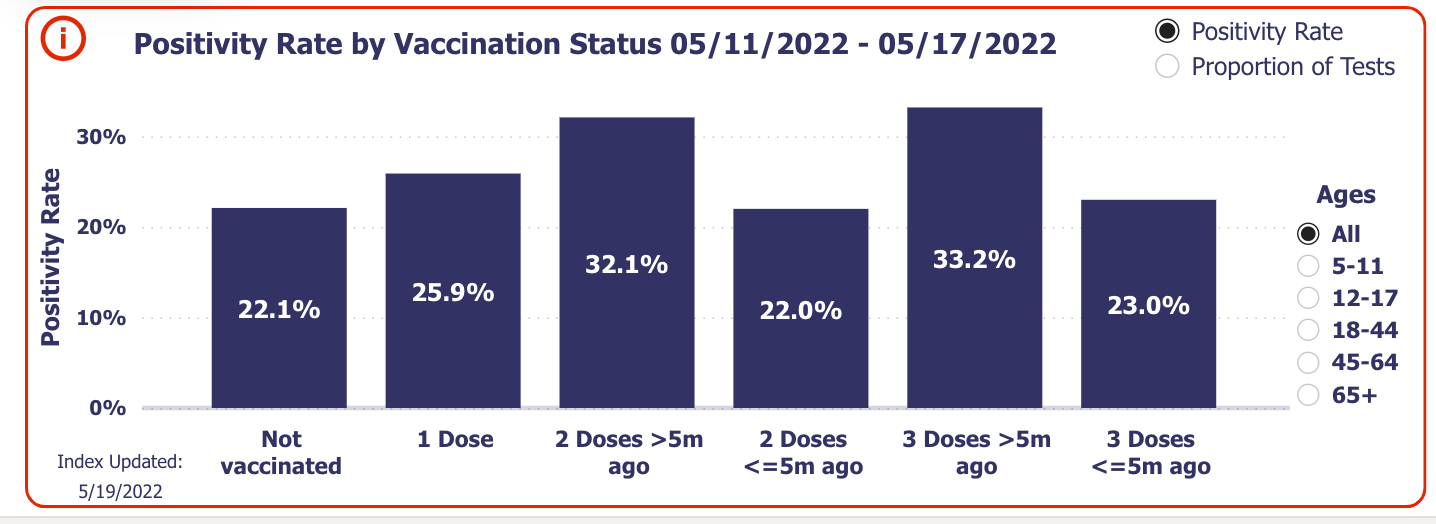
Walgreens, a recent source divorced from government-associated data collecting, shows that both two and three-dose vaccine recipients (greater than 5 months after their shots) are leading Covid case positivity rates in the U.S.
Much like the JAMA study, kids in the 5-11 and 12-17 age groups are showing those with a second dose greater than 5 months ago are leading the positive rate race.
Do we really want to expose kids to this possible fate? And for what purpose? There is no emergency in this age group. If anything, their natural immunity has acted like a barrier for the terrified laptop class and older, more vulnerable populations.







they're not even trying to hide it anymore, this is a deliberate attempt at a genocide, they know the "vaccines" are dangerous, and they don't care. This is a kill shot, plain and simple. Because what can we do about it when the ones in charge are the criminals? Who can bring them to justice?
It's a parent's responsibility to protect their child...we can pray for God's wisdom to do that. Parents keep sending their children to public indoctrination schools and then complain about what they are being taught!
HELLO!!!
My sister got her 3rd jab after all of the info I gave her, and after her grandson was diagnosed with Myocarditis!! Goes to show you how STUPID the left is!!!